Show me the code
library(readxl)
library(tidyverse)Copernicus institute of sustainable development, Utrecht University
Amsterdam sustainability institute, Vrije Universiteit Amsterdam
Departamento de Solos Centro de Ciências Agrárias, Universidade Federal de Vicosa, Brazil
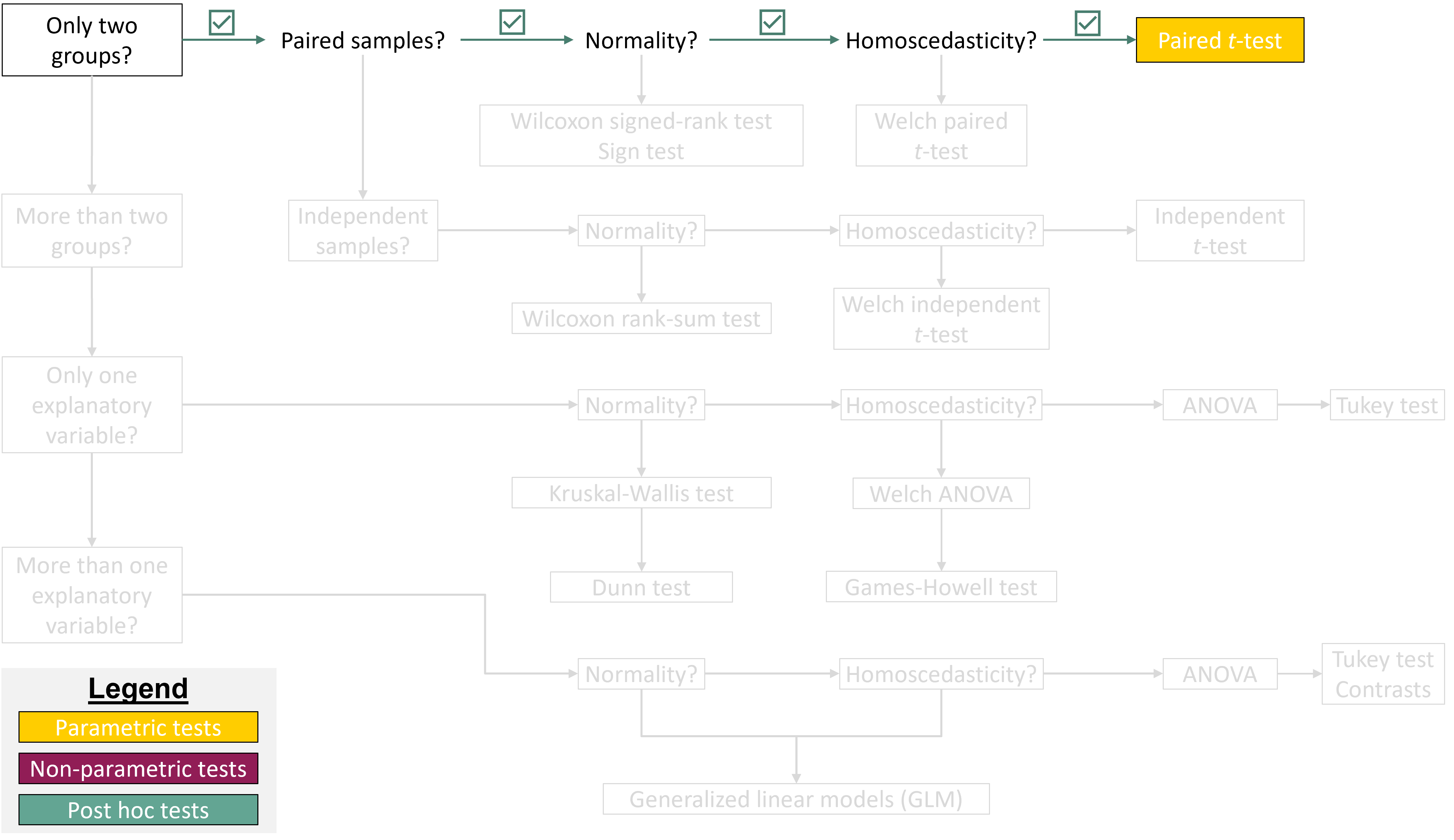
In this tutorial, we will enter the world of inferential statistics. We will use R to perform our first statistical test! By the end of this tutorial, you will be able to calculate z-scores and tail probabilities, check the normality assumption of the data, calculate 95% confidence intervals, perform a paired t-test, and interpret its results in R.
Let’s get started!
To work on this tutorial, you will need to load the readxl and tidyverse packages. You can load them using library().
Start by importing the data used in this course (see previous tutorial).
The tutorial dataset contains observations for 36 farms. The average coffee productivity measured across all farms was 1944.1 kg/ha/year and the standard deviation was 554.8 kg/ha/year. Assuming that coffee productivity data follow a normal distribution (we will explain later in this tutorial how to assess this assumption), the corresponding theoretical normal distribution is shown in Figure 1.
Now, suppose that we are interested in calculating the probability that a randomly selected farm has a coffee productivity greater or smaller than a specific productivity value. To answer such a question, we first need to calculate a z-score.
A z-score is a standardized measure that indicates how far a specific value or observation (\(y\)) is from the mean (\(\bar{y}\)) of a dataset, expressed in units of standard deviation (\(s\)). A positive z-score means the value is above the mean, while a negative z-score indicates it is below the mean.
\[ z = \frac{y - \bar{y}}{s}\ \tag{1}\]
If the original variable (Y) follows a normal distribution, the z-scores calculated from this variable will follow a standard normal distribution (mean of zero and standard deviation of one) (Figure 2).
Using Equation 1, calculate the z-score for all coffee productivity values (coffee_prod variable).
Suppose that we want to calculate the probability that a randomly selected farm has a coffee productivity greater than 2500 kg/ha/year. To answer this question, we first need to convert this value into a z-score (Equation 1).
We now need to calculate the probability of a value of the standard normal taking a value at least as large as the calculated z-score (1.002). Graphically, this corresponds to the area under the curve highlighted in yellow in Figure 3. In R, this probability can be obtained using the function pnorm(). You can also calculate this probability using the table of z values provided on Brightspace. Make sure you can use both options for the exam.
The pnorm() function takes four main arguments:
q: a z-score value (in our case: 1.002)
mean: the mean value of the normal distribution (should be zero for the standard normal, which is the default value)
sd: the standard deviation of the normal distribution (should be one for the standard normal, which is the default value)
lower.tail: a logical argument (can be either TRUE or FALSE). If set to TRUE, probabilities are \(P(X \le x)\). If set to FALSE, probabilities are \(P(X > x)\) (Figure 4)
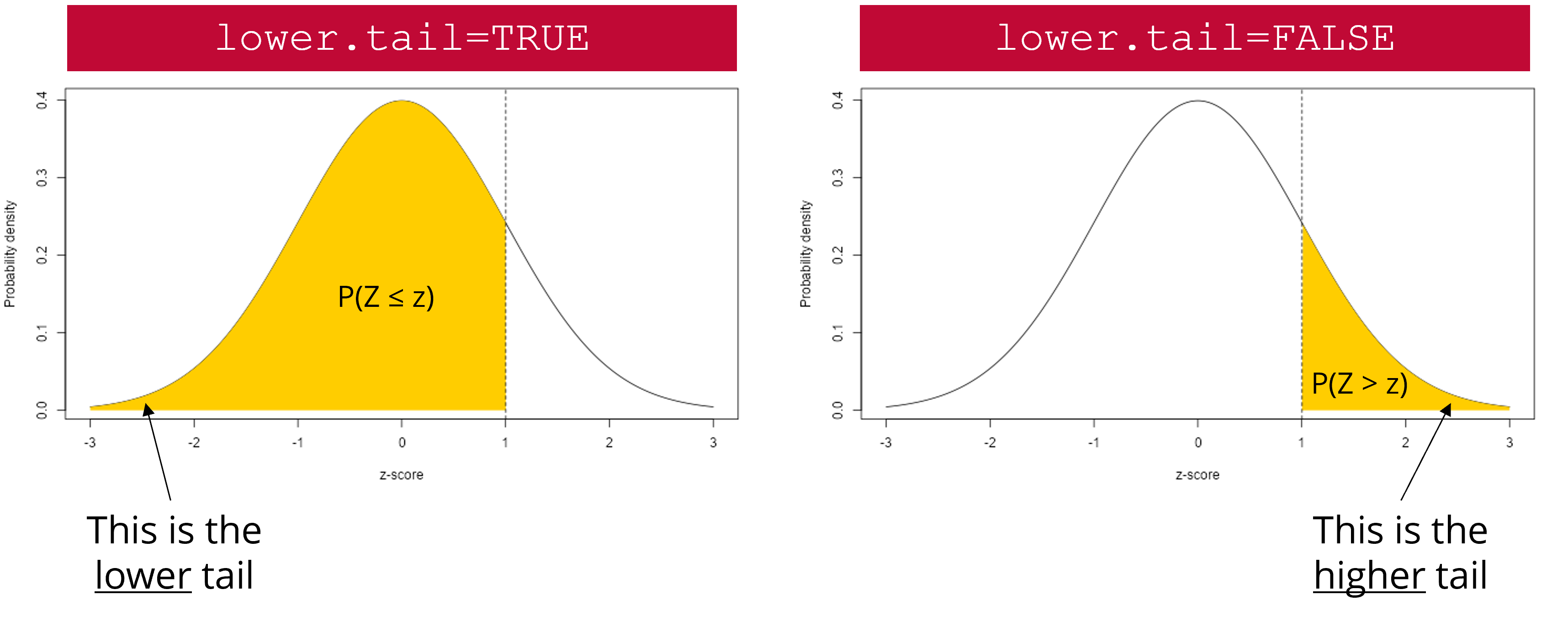
lower.tail argument works. The yellow area represents the probability that will be returned by pnorm() if lower.tail is set to TRUE (left scenario) or FALSE (right scenario).
Calculate the probability that a randomly selected farm has a coffee productivity greater than 2500 kg/ha/year. Do this using pnorm() and the table of z values provided on Brightspace.
Calculate the probability that a randomly selected farm has a coffee productivity comprised between 800 and 1200 kg/ha/year. Graphically, this means that you need to calculate the area under the curve highlighted in yellow in Figure 5. Do this using pnorm() and the table of z values provided on Brightspace.
#Calculate z-scores
low_z <- (800 - mean(data$coffee_prod))/sd(data$coffee_prod)
high_z <- (1200 - mean(data$coffee_prod))/sd(data$coffee_prod)
#Calculate tail probabilities
p_low_z <- pnorm(low_z, lower.tail = TRUE)
p_high_z <- pnorm(high_z, lower.tail = TRUE)
#Calculate the probability that a randomly selected farm has a coffee productivity comprised between 800 and 1200 kg/ha/year
p_high_z - p_low_zThe probability that a randomly selected farm has a coffee productivity comprised between 800 and 1200 kg/ha/year is 7%.
Many statistical tests, including t-tests, assume that the data are normally distributed within each group being compared. It is therefore important to check this assumption before conducting a statistical test. If your data do not follow a normal distribution, there are several approaches you can take. One option is to use a non-parametric test or model that does not assume normality (this will be discussed later in this course). Another option is to transform the data to make it more normally distributed, for example, using a log, or square root transformation, before applying a parametric test.
Normality can be assessed using both formal statistical tests, such as the Shapiro-Wilk test, and graphical tools, such as histograms and Q–Q plots. It is often best to use them in combination because the outcome of Shapiro-Wilk’s test is sensitive to sample size. With small sample sizes, the test has low statistical power, meaning it may fail to detect departures from normality even when the data are not necessarily normal. With large sample sizes, the test becomes extremely sensitive, so even very small deviations from normality can lead to a significant result.
In the Shapiro-Wilk’s test, the null hypothesis is that the data follow a normal distribution, while the alternative hypothesis is that they do not. Note that this test requires a sufficient sample size to be reliable (20-30 observations).
In R, a Shapiro-Wilk’s normality test can be carried out using shapiro.test(). The only argument to provide is a vector of data values (which may contain missing values).
Use the Shapiro-Wilk’s test to check that coffee productivity data (coffee_prod variable) is normally distributed within each farm type (farm_type variable). What can you conclude from these tests?
For all tests, the calculated p-value is much greater than 0.05. Therefore, we fail to reject the null hypothesis that coffee production data follow a normal distribution within each farm type.
Given the small number of coffee productivity observations available for each farm type in our dataset, creating histograms (one per farm type) is not very informative at this stage. Make sure you understand how these graphs are constructed and how to interpret them using the content of the lectures. We will revisit histograms later in the course, once we are more familiar with the concept of residuals.
Another important graphical tool to assess whether data follow a normal distribution is the Q-Q plot, short for Quantile-Quantile plot. A Q-Q plot compares the quantiles of the observed data to the quantiles expected under a normal distribution. If the data are approximately normally distributed, the points in the plot will lie close to a straight diagonal line. Systematic deviations from this line, such as curvature or extreme points at the ends, would indicate departures from normality.
In base R, you can create Q-Q plots by combining the functions qqnorm() and qqline(). However, since we need to create a Q-Q plot separately for each farm type, it is easier and faster to use ggplot2 functions to do this. In ggplot2, Q-Q plots are created using stat_qq() to plot the sample quantiles against the theoretical quantiles of a normal distribution, and stat_qq_line() to add a reference line. The function stat_qq() requires the sample argument, which specifies the variable for which the quantiles are to be computed and compared to the theoretical distribution (in our case: coffee_prod).
In our example, where we compare coffee productivity among farms, ggplot2 makes it easy to create a separate Q-Q plot for each farm type using faceting, without writing much additional code. Faceting can be done with facet_wrap() or facet_grid(), allowing you to visualize a Q-Q plot for each farm type side by side.
facet_grid() forms a matrix of panels defined by row and column variables. It is particularly useful when you have two discrete variables and all variable combinations exist in the data.
facet_wrap() is generally used when you want to create figure panels based on a single discrete variable (which is the case in this example).
Create Q-Q plots to check that coffee productivity data (coffee_prod variable) is normally distributed within each farm type (farm_type variable). What can you conclude from these graphs?
For Eco and Large farms, the points lie closer to the reference line than for Con farms (with a few exceptions), indicating that coffee productivity data from Con farms may deviate more from normality than data from the other farm types. However, these patterns should be interpreted with caution, as the sample size is small (12 observations per farm).
A 95% confidence interval (95% CI) is a range of values that is intended to contain the true population parameter (such as the mean value of the population) with a specified level of confidence (here: 95%). Its meaning is based on repeated sampling: if we were to take 100 random samples from the same population and compute a 95% confidence interval for each sample, approximately 95 of those intervals would include the true population mean.
When sample size is small (\(n<30\)), confidence intervals are calculated using quantile values (also referred to as critical values) of Student’s t distribution with \(n-1\) degrees of freedom (Equation 2). As the sample size increases, the t distribution converges to the standard normal distribution. For larger sample sizes (\(n>30\)), the critical values from the t distribution are very close to those from the normal distribution, so confidence intervals based on either distribution will be nearly identical.
Confidence intervals are always two-tailed because our estimate of the population mean (which is the sample mean, \(\bar{y}\)) may be larger or smaller than the true population mean (\(\mu\)), which is often unknown. To calculate a 95% CI, we therefore need to calculate the quantile value of Student’s t distribution associated with \(\alpha=0.05/2=0.025\). In R, this can be done with the qt() function.
\[ \bar{y} \pm t_{0.975,\,n-1} \cdot \frac{s}{\sqrt{n}} \tag{2}\]
The qt() function takes three main arguments:
p: a vector of probabilities (for a 95% CI, it will be 0.975)
df: the number of degrees of freedom (for a 95% CI, it will be \(n-1\))
lower.tail: a logical argument (can be either TRUE or FALSE). If set to TRUE, probabilities are \(P(X \le x)\). If set to FALSE, probabilities are \(P(X > x)\)
Calculate the 95% confidence interval for coffee productivity (coffee_prod variable) for each farm type. Do this both manually (using the table of critical values of Student’s t distribution available on Brightspace) as well as using the qt() function in R. Make sure you are able to use both methods for the exam.
#Calculate the number of observations for each farm type
n_con <- nrow(dplyr::filter(data, farm_type == "con"))
n_eco <- nrow(dplyr::filter(data, farm_type == "eco"))
n_large <- nrow(dplyr::filter(data, farm_type == "large"))
#Calculate the mean value of coffee productivity in each farm type
mean_con <- mean(dplyr::filter(data, farm_type == "con")$coffee_prod)
mean_eco <- mean(dplyr::filter(data, farm_type == "eco")$coffee_prod)
mean_large <- mean(dplyr::filter(data, farm_type == "large")$coffee_prod)
#Calculate the standard deviation value of coffee productivity in each farm type
sd_con <- sd(dplyr::filter(data, farm_type == "con")$coffee_prod)
sd_eco <- sd(dplyr::filter(data, farm_type == "eco")$coffee_prod)
sd_large <- sd(dplyr::filter(data, farm_type == "large")$coffee_prod)
#Calculate lower limits of 95% CI for each farm type
low_CI_con <- mean_con - qt(0.975, n_con-1)*sd_con/sqrt(n_con)
low_CI_eco <- mean_eco - qt(0.975, n_eco-1)*sd_eco/sqrt(n_eco)
low_CI_large <- mean_large - qt(0.975, n_large-1)*sd_large/sqrt(n_large)
#Calculate upper limits of 95% CI for each farm type
up_CI_con <- mean_con + qt(0.975, n_con-1)*sd_con/sqrt(n_con)
up_CI_eco <- mean_eco + qt(0.975, n_eco-1)*sd_eco/sqrt(n_eco)
up_CI_large <- mean_large + qt(0.975, n_large-1)*sd_large/sqrt(n_large)95% confidence interval for Con farms: [1686.9-2449.4] kg/ha/year
95% confidence interval for Eco farms: [1563.9-2116.5] kg/ha/year
95% confidence interval for Large farms: [1521.6-2326.5] kg/ha/year
Standard deviation describes how much individual observations in a dataset differ from the average. A large standard deviation means the data points are widely spread out, while a small one means they cluster closely around the mean.
Standard error is the standard deviation of the sampling distribution of the mean. It is mathematically related to the standard deviation. It measures how variable the sample mean will be, if you repeat the whole study many times (e.g., if you get a new random sample from the same population and calculate the mean value of that sample). A smaller standard error means the sample mean is likely to be closer to the true population mean, while a larger one indicates more uncertainty.
A 95% confidence interval gives a range of values that is likely to contain the true population mean (with 95% confidence), based on the sample data. It is mathematically related to the standard error. It means that if the same study were repeated many times, about 95% of the calculated intervals would contain the true population mean. A narrow interval indicates a precise estimate, while a wide interval indicates greater uncertainty.
We want to determine whether coffee productivity measured in 2020 (after an intervention from extension services) is higher than that measured in 2015 (before an intervention from extension services). In other words, did the intervention increase coffee productivity on farms?
In this example, the samples are paired because coffee productivity was measured on the same farms in two different years (before and after an intervention). This comparison also involves two groups: productivity measured in 2015 versus productivity measured in 2020. To answer our research question, a paired t-test seems appropriate.
Based on the information above, write a null and an alternative hypothesis for the paired t-test. Is the hypothesis directed or undirected?
The hypotheses for this test would be as follows:
Null hypothesis: there is no difference in coffee productivity between 2015 and 2020.
Alternative hypothesis: coffee productivity measured in 2020 is higher than coffee productivity measured in 2025 (which is what we would expect if the intervention had been successful).
This is an example of a directed hypothesis (right-tailed), because we expect an increase in productivity following the intervention.
Before running the test, we must first check whether our data meet the assumptions of a paired t-test (normality and homoscedasticity).
Earlier in this tutorial, you learned about methods for checking whether data follow a normal distribution (Shapiro-Wilk’s test and Q-Q plots). We can use the same methods here, but first we need to calculate the differences in coffee productivity between 2020 and 2015 for each farm (because we want to perform a paired t-test). We can then check whether these differences follow a normal distribution.
coffee_prod variable) and 2015 (coffee_prod_past variable). Store these differences in a new column called coffee_prod_diff in data.The p-value of Shapiro-Wilk’s test is higher than the significance level (0.05). Therefore, we fail to reject the null hypothesis and can reasonably assume that the data are normally distributed. The Q-Q plot suggests a slight skew in the data, but because the Shapiro-Wilk test is non-significant and most points lie close to the reference line, the normality assumption is considered acceptable.
Does coffee productivity measured in 2015 and 2020 show equal variances?
To answer this question, we can use Bartlett’s test. The null hypothesis of this test is that the groups have equal variance, while the alternative hypothesis is that they have different variances.
In R, this test can be carried out using the bartlett.test() function, which requires two main arguments:
x: a numeric vector of data values
g: a vector or factor object giving the group for the corresponding elements of x
To run this test, we would therefore need to reorganize the data a bit, so that we can have a data frame with a column for x (the coffee productivity values) and a column for g (the groups: 2015 and 2020).
bartlett.test(). One way to do this is to create a new data frame (data_bartlett) using the function data.frame() to combine variables into a single data frame. This data frame needs to contain two columns: x and g (but you could also name them differently). The column x contains the response variable and is created by concatenating (c()) coffee productivity values from two different years (coffee_prod_past and coffee_prod). The column g defines a grouping variable and is generated using rep(), which repeats the values 2015 and 2020 for each row in the original dataset (nrow(data)).The p-value of Bartlett’s test is larger than the significance threshold (0.05). Therefore, we fail to reject the null hypothesis that coffee productivity data measured in 2015 and 2020 have equal variances.
Now that we know that our data satisfy the underlying assumptions of a paired t-test, we can carry out the statistical test.
First, let’s do this using the “slow way” by manually computing the test statistic (\(t\)) and the p-value of the test.
For a paired t-test, the test statistic (\(t\)) can be calculated using Equation 3, where \(\bar{v}\) is the average difference in coffee productivity, \(s_v\) is the standard deviation of the differences, and \(n\) is the number of paired observations.
\[ t = \frac{\bar{v}}{s_v / \sqrt{n}} \tag{3}\]
Under the null hypothesis (i.e., if there is no difference in the means of the two populations being compared), the \(t\) statistic follows a Student’s t distribution with \(n-1\) degrees of freedom, where \(n\) is the number of paired observations (Figure 6).
The value of the test statistic is 5.372
The critical value of the test is 1.691
The absolute value of the calculated test statistic (\(t\)) is greater than the critical value. Therefore, we reject the null hypothesis that there is no difference in coffee productivity between 2015 and 2020. We conclude that the intervention led to an increase in coffee productivity.
We can now calculate the p-value associated with this test. We can do this using the pt() function in R.
The p-value is not the probability that the null hypothesis of your test is true.
In this paired t-test, the p-value represents the probability of obtaining a \(t\) statistic at least as extreme as the one observed if the null hypothesis were true (i.e, if there were actually no difference in coffee productivity between 2015 and 2020). A small p-value indicates that the observed result would be unlikely to occur purely by chance under the null hypothesis. If the p-value is sufficiently small (typically below 0.05), we would reject the null hypothesis in favor of the alternative hypothesis.
To calculate this p-value, it is important to remember that we are testing a directed (one-sided) hypothesis, as we expect coffee productivity in 2020 to be higher than in 2015. Graphically, this corresponds to calculating the area under the distribution curve to the right of the dashed vertical line shown in Figure 6. This area represents the p-value and appears to be very small.
In R, this probability can be calculated using the function pt(), which takes three main arguments:
q: a vector of quantiles (in this example, it is the value of the observed t statistic)
df: the number of degrees of freedom (\(n-1\))
lower.tail: a logical argument (can be either TRUE or FALSE). If set to TRUE, probabilities are \(P(X \le x)\). If set to FALSE, probabilities are \(P(X > x)\)
Calculate the p-value of the paired t-test using pt(). What can you conclude from this test?
The calculated p-value is 2.6024076^{-6}, which is much lower than the significance threshold (0.05). Therefore, we reject the null hypothesis that there is no difference in coffee productivity between 2015 and 2020. We conclude that the intervention led to an increase in coffee productivity. On average, coffee productivity in 2020 increased by 263.9 kg/ha/year.
Of course, t-tests can be easily performed in R using the t.test() function. Here are the most important arguments of this function:
x and y: the numeric vectors of data to compare (one vector per group)
paired: a logical value (TRUE or FALSE) indicating whether the samples are paired (TRUE) or independent (FALSE)
alternative: specifies the type of test; "two.sided" (default, for undirected hypotheses), "greater", or "less"
mu: the expected difference between group means if the null hypothesis is true
var.equal: a logical variable indicating whether to treat the two variances as being equal. If TRUE then the pooled variance is used to estimate the variance otherwise the Welch (or Satterthwaite) approximation to the degrees of freedom is used.
conf.level: the confidence level for the interval (default is 0.95)
Use the t.test() function to perform the statistical test. What information is shown in the R output? What can you conclude from this test? Write a few sentences to report this result, including the test statistic, p-value, sample size (degrees of freedom), and interpretation.
Paired t-test
data: data$coffee_prod and data$coffee_prod_past
t = 5.3716, df = 35, p-value = 2.602e-06
alternative hypothesis: true mean difference is greater than 0
95 percent confidence interval:
180.8656 Inf
sample estimates:
mean difference
263.8594 We conducted a paired t-test to assess if an intervention from extension services resulted in increased coffee productivity (Figure 7). Therefore, coffee productivity was measured in the same farms before and after the intervention (n=36 farms). The difference between coffee productivity before and after the intervention was significant (t= 5.37; p-value < 0.001) indicating that the intervention had a positive effect. On average, coffee productivity in 2020 was 15.7% greater than in 2015.
---
title: "Tutorial 3: Comparing two groups (part 1)"
author:
- name: Benjamin Delory
orcid: 0000-0002-1190-8060
email: b.m.m.delory@uu.nl
affiliations:
- name: Copernicus institute of sustainable development, Utrecht University
- name: Natalie Davis
orcid: 0000-0002-2678-0389
email: n.a.davis@vu.nl
affiliations:
- name: Amsterdam sustainability institute, Vrije Universiteit Amsterdam
- name: Heitor Mancini Teixeira
orcid: 0000-0001-6992-0671
email: heitor.teixeira@ufv.br
affiliations:
- name: Departamento de Solos Centro de Ciências Agrárias, Universidade Federal de Vicosa, Brazil
format: html
editor: visual
editor_options:
chunk_output_type: console
image: /Images/Rlogo.png
---
## About this tutorial
In this tutorial, we will enter the world of inferential statistics. We will use R to perform our first statistical test! By the end of this tutorial, you will be able to calculate z-scores and tail probabilities, check the normality assumption of the data, calculate 95% confidence intervals, perform a paired t-test, and interpret its results in R.
Let's get started!
## Where are we in the course?
{fig-align="center"}
## Load R packages
To work on this tutorial, you will need to load the *readxl* and *tidyverse* packages. You can load them using `library()`.
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
library(readxl)
library(tidyverse)
```
## Importing data
Start by importing the data used in this course (see previous tutorial).
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
#Option 1 (csv file)
data <- read_csv("gss_statistics_master_data_set2.csv")
#Option 2 (Excel file)
data <- read_excel("gss_statistics_master_data_set2.xlsx")
```
## z-scores and tail probabilities
### Theoretical normal distribution of coffee productivity data
The tutorial dataset contains observations for 36 farms. The average coffee productivity measured across all farms was `{r} round(mean(data$coffee_prod), 1)` kg/ha/year and the standard deviation was `{r} round(sd(data$coffee_prod), 1)` kg/ha/year. Assuming that coffee productivity data follow a normal distribution (we will explain later in this tutorial how to assess this assumption), the corresponding theoretical normal distribution is shown in @fig-theoreticalnormalcoffee.
```{r}
#| eval: true
#| echo: false
#| warning: false
#| message: false
#| fig-width: 6
#| fig-height: 6
#| fig-align: center
#| fig-cap: "Theoretical normal distribution of coffee productivity"
#| label: fig-theoreticalnormalcoffee
ht <- seq(500,3500,1)
plot(ht,dnorm(ht,mean(data$coffee_prod),sd(data$coffee_prod)),type="l",col="black", ylab="Probability density",xlab="Coffee productivity (kg/ha/year)")
```
Now, suppose that we are interested in calculating the probability that a randomly selected farm has a coffee productivity greater or smaller than a specific productivity value. To answer such a question, we first need to calculate a z-score.
### Calculating z-scores
A z-score is a standardized measure that indicates how far a specific value or observation ($y$) is from the mean ($\bar{y}$) of a dataset, expressed in units of standard deviation ($s$). A positive z-score means the value is above the mean, while a negative z-score indicates it is below the mean.
$$
z = \frac{y - \bar{y}}{s}\
$$ {#eq-zscore}
If the original variable (Y) follows a normal distribution, the z-scores calculated from this variable will follow a **standard normal distribution** (mean of zero and standard deviation of one) (@fig-standardnormal).
```{r}
#| eval: true
#| echo: false
#| warning: false
#| message: false
#| fig-width: 6
#| fig-height: 6
#| fig-align: center
#| fig-cap: "Standard normal distribution"
#| label: fig-standardnormal
ht <- seq(-3,3,0.01)
plot(ht,dnorm(ht,0,1),type="l",col="black", ylab="Probability density",xlab="z-score")
```
::: callout-tip
## Exercise 1
Using @eq-zscore, calculate the z-score for all coffee productivity values (`coffee_prod` variable).
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
zscores <- (data$coffee_prod - mean(data$coffee_prod))/sd(data$coffee_prod)
```
:::
### Calculating tail probabilities
Suppose that we want to calculate the probability that a randomly selected farm has a coffee productivity greater than 2500 kg/ha/year. To answer this question, we first need to convert this value into a z-score (@eq-zscore).
::: callout-tip
## Exercise 2
Calculate the z-score for a coffee productivity of 2500 kg/ha/year.
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
zscore <- (2500 - mean(data$coffee_prod))/sd(data$coffee_prod)
```
:::
We now need to calculate the probability of a value of the standard normal taking a value at least as large as the calculated z-score (`{r} round(zscore, 3)`). Graphically, this corresponds to the area under the curve highlighted in yellow in @fig-standardnormalwitharea1. In R, this probability can be obtained using the function `pnorm()`. You can also calculate this probability using the table of z values provided on Brightspace. [**Make sure you can use both options for the exam**]{.underline}.
```{r}
#| eval: true
#| echo: false
#| warning: false
#| message: false
#| fig-width: 6
#| fig-height: 6
#| fig-align: center
#| fig-cap: "Standard normal distribution. The area under the curve highlighted in yellow is the probability of a value of the standard normal taking a value at least as large as the calculated z-score."
#| label: fig-standardnormalwitharea1
ht <- seq(-3,3,0.01)
pd <- dnorm(ht,0,1)
plot(ht,dnorm(ht,0,1),type="l",col="black", ylab="Probability density", xlab="z-score", main="Standard normal distribution")
xv <- ht[ht>=zscore]
yv <- pd[ht>=zscore]
xv <- c(xv,3,zscore)
yv <- c(yv,0,0)
polygon(xv,yv,col="#FFCD00",border=NA)
abline(v=zscore, lty=2)
```
The `pnorm()` function takes four main arguments:
- `q`: a z-score value (in our case: `{r} round(zscore, 3)`)
- `mean`: the mean value of the normal distribution (should be zero for the standard normal, which is the default value)
- `sd`: the standard deviation of the normal distribution (should be one for the standard normal, which is the default value)
- `lower.tail`: a logical argument (can be either `TRUE` or `FALSE`). If set to `TRUE`, probabilities are $P(X \le x)$. If set to `FALSE`, probabilities are $P(X > x)$ (@fig-lowertail)
{#fig-lowertail fig-align="center"}
:::: callout-tip
## Exercise 3
Calculate the probability that a randomly selected farm has a coffee productivity greater than 2500 kg/ha/year. Do this using `pnorm()` and the table of z values provided on Brightspace.
::: panel-tabset
## Code
```{r}
#| eval: true
#| echo: true
#| warning: false
#| message: false
#| output: false
#Option 1
pnorm(zscore, lower.tail = FALSE)
#Option 2
1-pnorm(zscore, lower.tail = TRUE)
```
## Answer
The probability that a randomly selected farm has a coffee productivity greater than 2500 kg/ha/year is `{r} round(100*pnorm(zscore, lower.tail = FALSE), 1)`%.
:::
::::
### Challenge to do at home
:::: callout-tip
## Exercise 4
Calculate the probability that a randomly selected farm has a coffee productivity comprised between 800 and 1200 kg/ha/year. Graphically, this means that you need to calculate the area under the curve highlighted in yellow in @fig-theoreticalnormalwitharea1. Do this using `pnorm()` and the table of z values provided on Brightspace.
```{r}
#| eval: true
#| echo: false
#| warning: false
#| message: false
#| fig-width: 6
#| fig-height: 6
#| fig-align: center
#| fig-cap: "Theoretical normal distribution of coffee productivity. The area under the curve highlighted in yellow is the probability that a randomly selected farm has a coffee productivity comprised between 800 and 1200 kg/ha/year."
#| label: fig-theoreticalnormalwitharea1
low_z <- (800 - mean(data$coffee_prod))/sd(data$coffee_prod)
high_z <- (1200 - mean(data$coffee_prod))/sd(data$coffee_prod)
ht <- seq(500,3500,1)
pd <- dnorm(ht,mean(data$coffee_prod),sd(data$coffee_prod))
plot(ht,dnorm(ht,mean(data$coffee_prod),sd(data$coffee_prod)),type="l", col="black", ylab="Probability density", xlab="Coffee productivity (kg/ha/year)", main="Theoretical normal distribution of coffee productivity")
xv <- ht[ht>=800 & ht <= 1200]
yv <- pd[ht>=800 & ht <= 1200]
xv <- c(xv,1200,800)
yv <- c(yv,0,0)
polygon(xv,yv,col="#FFCD00",border=NA)
```
::: panel-tabset
## Code
```{r}
#| eval: true
#| echo: true
#| warning: false
#| message: false
#| output: false
#Calculate z-scores
low_z <- (800 - mean(data$coffee_prod))/sd(data$coffee_prod)
high_z <- (1200 - mean(data$coffee_prod))/sd(data$coffee_prod)
#Calculate tail probabilities
p_low_z <- pnorm(low_z, lower.tail = TRUE)
p_high_z <- pnorm(high_z, lower.tail = TRUE)
#Calculate the probability that a randomly selected farm has a coffee productivity comprised between 800 and 1200 kg/ha/year
p_high_z - p_low_z
```
## Answer
The probability that a randomly selected farm has a coffee productivity comprised between 800 and 1200 kg/ha/year is `{r} round(100*(p_high_z - p_low_z), 1)`%.
:::
::::
## Checking for data normality
Many statistical tests, including *t*-tests, assume that the data are normally distributed within each group being compared. It is therefore important to check this assumption before conducting a statistical test. If your data do not follow a normal distribution, there are several approaches you can take. One option is to use a non-parametric test or model that does not assume normality (this will be discussed later in this course). Another option is to transform the data to make it more normally distributed, for example, using a log, or square root transformation, before applying a parametric test.
Normality can be assessed using both formal statistical tests, such as the Shapiro-Wilk test, and graphical tools, such as histograms and Q–Q plots. It is often best to use them in combination because the outcome of Shapiro-Wilk's test is sensitive to sample size. With small sample sizes, the test has low statistical power, meaning it may fail to detect departures from normality even when the data are not necessarily normal. With large sample sizes, the test becomes extremely sensitive, so even very small deviations from normality can lead to a significant result.
### Shapiro-Wilk's test
In the Shapiro-Wilk's test, the null hypothesis is that the data follow a normal distribution, while the alternative hypothesis is that they do not. Note that this test requires a sufficient sample size to be reliable (20-30 observations).
In R, a Shapiro-Wilk's normality test can be carried out using `shapiro.test()`. The only argument to provide is a vector of data values (which may contain missing values).
:::: callout-tip
## Exercise 5
Use the Shapiro-Wilk's test to check that coffee productivity data (`coffee_prod` variable) is normally distributed within each farm type (`farm_type` variable). What can you conclude from these tests?
::: panel-tabset
## Con farms
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
#Filter data for con farms
data_con <- dplyr::filter(data, farm_type == "con")
#Shapiro-Wilk's test
shapiro.test(data_con$coffee_prod)
```
## Eco farms
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
#Filter data for eco farms
data_eco <- dplyr::filter(data, farm_type == "eco")
#Shapiro-Wilk's test
shapiro.test(data_eco$coffee_prod)
```
## Large farms
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
#Filter data for con farms
data_large <- dplyr::filter(data, farm_type == "large")
#Shapiro-Wilk's test
shapiro.test(data_large$coffee_prod)
```
## Conclusion from tests
For all tests, the calculated p-value is much greater than 0.05. Therefore, we fail to reject the null hypothesis that coffee production data follow a normal distribution within each farm type.
:::
::::
### Q-Q plots
Given the small number of coffee productivity observations available for each farm type in our dataset, creating histograms (one per farm type) is not very informative at this stage. [**Make sure you understand how these graphs are constructed and how to interpret them using the content of the lectures**]{.underline}. We will revisit histograms later in the course, once we are more familiar with the concept of residuals.
Another important graphical tool to assess whether data follow a normal distribution is the Q-Q plot, short for Quantile-Quantile plot. A Q-Q plot compares the quantiles of the observed data to the quantiles expected under a normal distribution. If the data are approximately normally distributed, the points in the plot will lie close to a straight diagonal line. Systematic deviations from this line, such as curvature or extreme points at the ends, would indicate departures from normality.
In base R, you can create Q-Q plots by combining the functions `qqnorm()` and `qqline()`. However, since we need to create a Q-Q plot separately for each farm type, it is easier and faster to use *ggplot2* functions to do this. In *ggplot2*, Q-Q plots are created using `stat_qq()` to plot the sample quantiles against the theoretical quantiles of a normal distribution, and `stat_qq_line()` to add a reference line. The function `stat_qq()` requires the `sample` argument, which specifies the variable for which the quantiles are to be computed and compared to the theoretical distribution (in our case: `coffee_prod`).
In our example, where we compare coffee productivity among farms, *ggplot2* makes it easy to create a separate Q-Q plot for each farm type using faceting, without writing much additional code. Faceting can be done with `facet_wrap()` or `facet_grid()`, allowing you to visualize a Q-Q plot for each farm type side by side.
`facet_grid()` forms a matrix of panels defined by row and column variables. It is particularly useful when you have two discrete variables and all variable combinations exist in the data.
`facet_wrap()` is generally used when you want to create figure panels based on a single discrete variable (which is the case in this example).
:::: callout-tip
## Exercise 6
Create Q-Q plots to check that coffee productivity data (`coffee_prod` variable) is normally distributed within each farm type (`farm_type` variable). What can you conclude from these graphs?
::: panel-tabset
## Code
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
#| fig-align: center
#| fig-height: 4
ggplot(data, aes(sample = coffee_prod)) +
stat_qq() +
stat_qq_line() +
facet_wrap(~ farm_type) +
theme_bw() +
xlab("Theoretical quantiles")+
ylab("Sample quantiles")
```
## Answer
For Eco and Large farms, the points lie closer to the reference line than for Con farms (with a few exceptions), indicating that coffee productivity data from Con farms may deviate more from normality than data from the other farm types. However, these patterns should be interpreted with caution, as the sample size is small (12 observations per farm).
:::
::::
## Calculating 95% confidence intervals
A 95% confidence interval (95% CI) is a range of values that is intended to contain the true population parameter (such as the mean value of the population) with a specified level of confidence (here: 95%). Its meaning is based on repeated sampling: if we were to take 100 random samples from the same population and compute a 95% confidence interval for each sample, approximately 95 of those intervals would include the true population mean.
When sample size is small ($n<30$), confidence intervals are calculated using quantile values (also referred to as critical values) of Student's *t* distribution with $n-1$ degrees of freedom (@eq-confidenceinterval_t). As the sample size increases, the *t* distribution converges to the standard normal distribution. For larger sample sizes ($n>30$), the critical values from the *t* distribution are very close to those from the normal distribution, so confidence intervals based on either distribution will be nearly identical.
Confidence intervals are always two-tailed because our estimate of the population mean (which is the sample mean, $\bar{y}$) may be larger or smaller than the true population mean ($\mu$), which is often unknown. To calculate a 95% CI, we therefore need to calculate the quantile value of Student's *t* distribution associated with $\alpha=0.05/2=0.025$. In R, this can be done with the `qt()` function.
$$
\bar{y} \pm t_{0.975,\,n-1} \cdot \frac{s}{\sqrt{n}}
$$ {#eq-confidenceinterval_t}
The `qt()` function takes three main arguments:
- `p`: a vector of probabilities (for a 95% CI, it will be 0.975)
- `df`: the number of degrees of freedom (for a 95% CI, it will be $n-1$)
- `lower.tail`: a logical argument (can be either `TRUE` or `FALSE`). If set to `TRUE`, probabilities are $P(X \le x)$. If set to `FALSE`, probabilities are $P(X > x)$
:::: callout-tip
## Exercise 7
Calculate the 95% confidence interval for coffee productivity (`coffee_prod` variable) for each farm type. Do this both manually (using the table of critical values of Student's *t* distribution available on Brightspace) as well as using the `qt()` function in R. [**Make sure you are able to use both methods for the exam**]{.underline}.
::: panel-tabset
## Code
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
#Calculate the number of observations for each farm type
n_con <- nrow(dplyr::filter(data, farm_type == "con"))
n_eco <- nrow(dplyr::filter(data, farm_type == "eco"))
n_large <- nrow(dplyr::filter(data, farm_type == "large"))
#Calculate the mean value of coffee productivity in each farm type
mean_con <- mean(dplyr::filter(data, farm_type == "con")$coffee_prod)
mean_eco <- mean(dplyr::filter(data, farm_type == "eco")$coffee_prod)
mean_large <- mean(dplyr::filter(data, farm_type == "large")$coffee_prod)
#Calculate the standard deviation value of coffee productivity in each farm type
sd_con <- sd(dplyr::filter(data, farm_type == "con")$coffee_prod)
sd_eco <- sd(dplyr::filter(data, farm_type == "eco")$coffee_prod)
sd_large <- sd(dplyr::filter(data, farm_type == "large")$coffee_prod)
#Calculate lower limits of 95% CI for each farm type
low_CI_con <- mean_con - qt(0.975, n_con-1)*sd_con/sqrt(n_con)
low_CI_eco <- mean_eco - qt(0.975, n_eco-1)*sd_eco/sqrt(n_eco)
low_CI_large <- mean_large - qt(0.975, n_large-1)*sd_large/sqrt(n_large)
#Calculate upper limits of 95% CI for each farm type
up_CI_con <- mean_con + qt(0.975, n_con-1)*sd_con/sqrt(n_con)
up_CI_eco <- mean_eco + qt(0.975, n_eco-1)*sd_eco/sqrt(n_eco)
up_CI_large <- mean_large + qt(0.975, n_large-1)*sd_large/sqrt(n_large)
```
## Answer
95% confidence interval for Con farms: \[`{r} round(low_CI_con, 1)`-`{r} round(up_CI_con, 1)`\] kg/ha/year
95% confidence interval for Eco farms: \[`{r} round(low_CI_eco, 1)`-`{r} round(up_CI_eco, 1)`\] kg/ha/year
95% confidence interval for Large farms: \[`{r} round(low_CI_large, 1)`-`{r} round(up_CI_large, 1)`\] kg/ha/year
:::
::::
::: callout-note
## Standard deviation, standard error, and 95% confidence interval
**Standard deviation** describes how much individual observations in a dataset differ from the average. A large standard deviation means the data points are widely spread out, while a small one means they cluster closely around the mean.
**Standard error** is the standard deviation of the sampling distribution of the mean. It is mathematically related to the standard deviation. It measures how variable the sample mean will be, if you repeat the whole study many times (e.g., if you get a new random sample from the same population and calculate the mean value of that sample). A smaller standard error means the sample mean is likely to be closer to the true population mean, while a larger one indicates more uncertainty.
A **95% confidence interval** gives a range of values that is likely to contain the true population mean (with 95% confidence), based on the sample data. It is mathematically related to the standard error. It means that if the same study were repeated many times, about 95% of the calculated intervals would contain the true population mean. A narrow interval indicates a precise estimate, while a wide interval indicates greater uncertainty.
:::
## Doing a paired *t*-test
### Question and hypotheses
We want to determine whether coffee productivity measured in 2020 (after an intervention from extension services) is higher than that measured in 2015 (before an intervention from extension services). In other words, did the intervention increase coffee productivity on farms?
In this example, the samples are paired because coffee productivity was measured on the [same farms]{.underline} in two different years (before and after an intervention). This comparison also involves two groups: productivity measured in 2015 versus productivity measured in 2020. To answer our research question, a paired *t*-test seems appropriate.
:::: callout-tip
## Exercise 8
::: panel-tabset
## Question
Based on the information above, write a null and an alternative hypothesis for the paired *t*-test. Is the hypothesis directed or undirected?
## Answer
The hypotheses for this test would be as follows:
**Null hypothesis**: there is no difference in coffee productivity between 2015 and 2020.
**Alternative hypothesis**: coffee productivity measured in 2020 [is higher]{.underline} than coffee productivity measured in 2025 (which is what we would expect if the intervention had been successful).
This is an example of a directed hypothesis (right-tailed), because we expect an increase in productivity following the intervention.
:::
::::
Before running the test, we must first check whether our data meet the assumptions of a paired *t*-test (normality and homoscedasticity).
### Check assumptions
#### Normality
Earlier in this tutorial, you learned about methods for checking whether data follow a normal distribution (Shapiro-Wilk's test and Q-Q plots). We can use the same methods here, but first we need to calculate the differences in coffee productivity between 2020 and 2015 for each farm (because we want to perform a paired *t*-test). We can then check whether these differences follow a normal distribution.
:::: callout-tip
## Exercise 9
1. Calculate differences in coffee productivity between 2020 (`coffee_prod` variable) and 2015 (`coffee_prod_past` variable). Store these differences in a new column called `coffee_prod_diff` in `data`.
2. Use a Shapiro-Wilk's test to check whether differences in coffee productivity follow a normal distribution. here, you do not need to do this separately for each farm, because we want to make a comparison across farms.
3. Create a Q-Q plot to visually assess deviations from normality.
4. What can you conclude from the Shapiro-Wilk's test and the Q-Q plot?
::: panel-tabset
## Solution 1
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
data$coffee_prod_diff <- data$coffee_prod - data$coffee_prod_past
```
## Solution 2
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
shapiro.test(data$coffee_prod_diff)
```
## Solution 3
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
#| fig-align: center
#| fig-height: 5
#| fig-width: 5
ggplot(data, aes(sample = coffee_prod_diff)) +
stat_qq() +
stat_qq_line() +
theme_bw() +
xlab("Theoretical quantiles")+
ylab("Sample quantiles")
```
## Solution 4
The p-value of Shapiro-Wilk's test is higher than the significance level (0.05). Therefore, we fail to reject the null hypothesis and can reasonably assume that the data are normally distributed. The Q-Q plot suggests a slight skew in the data, but because the Shapiro-Wilk test is non-significant and most points lie close to the reference line, the normality assumption is considered acceptable.
:::
::::
#### Homoscedasticity
Does coffee productivity measured in 2015 and 2020 show equal variances?
To answer this question, we can use Bartlett's test. The null hypothesis of this test is that the groups have equal variance, while the alternative hypothesis is that they have different variances.
In R, this test can be carried out using the `bartlett.test()` function, which requires two main arguments:
- `x`: a numeric vector of data values
- `g`: a vector or factor object giving the group for the corresponding elements of `x`
To run this test, we would therefore need to reorganize the data a bit, so that we can have a data frame with a column for `x` (the coffee productivity values) and a column for `g` (the groups: 2015 and 2020).
:::: callout-tip
## Exercise 10
1. Reorganise the data so that it can be used in `bartlett.test()`. One way to do this is to create a new data frame (`data_bartlett`) using the function `data.frame()` to combine variables into a single data frame. This data frame needs to contain two columns: `x` and `g` (but you could also name them differently). The column `x` contains the response variable and is created by concatenating (`c()`) coffee productivity values from two different years (`coffee_prod_past` and `coffee_prod`). The column `g` defines a grouping variable and is generated using `rep()`, which repeats the values `2015` and `2020` for each row in the original dataset (`nrow(data)`).
2. Run the Bartlett's test.
3. What can you conclude from this test?
::: panel-tabset
## Solution 1
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
data_bartlett <- data.frame(x = c(data$coffee_prod_past, data$coffee_prod),
g = rep(c(2015, 2020), each=nrow(data)))
```
## Solution 2
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
bartlett.test(x = data_bartlett$x,
g = data_bartlett$g)
```
## Solution 3
The p-value of Bartlett's test is larger than the significance threshold (0.05). Therefore, we fail to reject the null hypothesis that coffee productivity data measured in 2015 and 2020 have equal variances.
:::
::::
### The slow way
Now that we know that our data satisfy the underlying assumptions of a paired *t*-test, we can carry out the statistical test.
First, let's do this using the "slow way" by manually computing the test statistic ($t$) and the p-value of the test.
For a paired *t*-test, the test statistic ($t$) can be calculated using @eq-pairedttest, where $\bar{v}$ is the average difference in coffee productivity, $s_v$ is the standard deviation of the differences, and $n$ is the number of paired observations.
$$
t = \frac{\bar{v}}{s_v / \sqrt{n}}
$$ {#eq-pairedttest}
Under the null hypothesis (i.e., if there is no difference in the means of the two populations being compared), the $t$ statistic follows a Student’s *t* distribution with $n-1$ degrees of freedom, where $n$ is the number of paired observations (@fig-studentdistribution).
:::: callout-tip
## Exercise 11
1. Calculate the value of the test statistic ($t$) using @eq-pairedttest.
2. Use the table of critical values of Student's *t* distribution available on Brightspace to obtain the critical value of the test. **Tip:** To find the critical value of a Student’s *t* distribution with 35 degrees of freedom, use linear interpolation between the tabulated values for 30 and 40 degrees of freedom. Because 35 lies exactly halfway between 30 and 40, this is equivalent to taking the average of the two critical values.
3. Based on the calculated test statistic and critical value, what can you conclude about the effect of the intervention on coffee productivity?
::: panel-tabset
## Solution 1
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
t <- mean(data$coffee_prod_diff)/(sd(data$coffee_prod_diff)/sqrt(nrow(data)))
```
The value of the test statistic is `{r} round(t, 3)`
## Solution 2
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
#35 degrees of freedom, directed hypothesis (5%)
#Equivalent to doing a linear interpolation
critical_value <- (1.697+1.684)/2
```
The critical value of the test is `{r} round(critical_value, 3)`
## Solution 3
The absolute value of the calculated test statistic ($t$) is greater than the critical value. Therefore, we reject the null hypothesis that there is no difference in coffee productivity between 2015 and 2020. We conclude that the intervention led to an increase in coffee productivity.
:::
::::
```{r}
#| eval: true
#| echo: false
#| warning: false
#| message: false
#| fig-width: 6
#| fig-height: 6
#| fig-align: center
#| fig-cap: "Student's t distribution with 35 degrees of freedom. The vertical dashed line shows the value of the observed t statistic (paired t-test)."
#| label: fig-studentdistribution
ht <- seq(-6,6,0.01)
pd <- dt(ht,df=35)
plot(ht,dt(ht,df=35),type="l",col="black", ylab="Probability density", xlab="t statistic")
xv <- ht[ht>=t]
yv <- pd[ht>=t]
xv <- c(xv,6,t)
yv <- c(yv,0,0)
polygon(xv,yv,col="#FFCD00",border=NA)
abline(v=t, lty=2)
```
We can now calculate the p-value associated with this test. We can do this using the `pt()` function in R.
::: callout-important
## What is (not) a p-value?
The p-value [**is not**]{.underline} the probability that the null hypothesis of your test is true.
In this paired t-test, the **p-value represents the probability of obtaining a** $t$ **statistic at least as extreme as the one observed if the null hypothesis were true** (i.e, if there were actually no difference in coffee productivity between 2015 and 2020). A small p-value indicates that the observed result would be unlikely to occur purely by chance under the null hypothesis. If the p-value is sufficiently small (typically below 0.05), we would reject the null hypothesis in favor of the alternative hypothesis.
:::
To calculate this p-value, it is important to remember that we are testing a directed (one-sided) hypothesis, as we expect coffee productivity in 2020 to be higher than in 2015. Graphically, this corresponds to calculating the area under the distribution curve to the right of the dashed vertical line shown in @fig-studentdistribution. This area represents the p-value and appears to be very small.
In R, this probability can be calculated using the function `pt()`, which takes three main arguments:
- `q`: a vector of quantiles (in this example, it is the value of the observed *t* statistic)
- `df`: the number of degrees of freedom ($n-1$)
- `lower.tail`: a logical argument (can be either `TRUE` or `FALSE`). If set to `TRUE`, probabilities are $P(X \le x)$. If set to `FALSE`, probabilities are $P(X > x)$
:::: callout-tip
## Exercise 12
Calculate the p-value of the paired *t*-test using `pt()`. What can you conclude from this test?
::: panel-tabset
## Code
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
p <- pt(t, df=nrow(data)-1, lower.tail=FALSE)
```
## Answer
The calculated p-value is `{r} p`, which is much lower than the significance threshold (0.05). Therefore, we reject the null hypothesis that there is no difference in coffee productivity between 2015 and 2020. We conclude that the intervention led to an increase in coffee productivity. On average, coffee productivity in 2020 increased by `{r} round(mean(data$coffee_prod_diff), 1)` kg/ha/year.
:::
::::
### The fast way
Of course, *t*-tests can be easily performed in R using the `t.test()` function. Here are the most important arguments of this function:
- `x` and `y`: the numeric vectors of data to compare (one vector per group)
- `paired`: a logical value (`TRUE` or `FALSE`) indicating whether the samples are paired (`TRUE`) or independent (`FALSE`)
- `alternative`: specifies the type of test; `"two.sided"` (default, for undirected hypotheses), `"greater"`, or `"less"`
- `mu`: the expected difference between group means if the null hypothesis is true
- `var.equal`: a logical variable indicating whether to treat the two variances as being equal. If `TRUE` then the pooled variance is used to estimate the variance otherwise the Welch (or Satterthwaite) approximation to the degrees of freedom is used.
- `conf.level`: the confidence level for the interval (default is 0.95)
:::: callout-tip
## Exercise 13
Use the `t.test()` function to perform the statistical test. What information is shown in the R output? What can you conclude from this test? Write a few sentences to report this result, including the test statistic, p-value, sample size (degrees of freedom), and interpretation.
::: panel-tabset
## Code
```{r}
#| eval: true
#| echo: true
#| message: false
#| warning: false
t.test(x = data$coffee_prod,
y = data$coffee_prod_past,
paired = TRUE,
alternative = "greater",
mu = 0,
var.equal = TRUE)
```
## Answer
We conducted a paired *t*-test to assess if an intervention from extension services resulted in increased coffee productivity (@fig-coffee-prod). Therefore, coffee productivity was measured in the same farms before and after the intervention (n=36 farms). The difference between coffee productivity before and after the intervention was significant (t= 5.37; p-value \< 0.001) indicating that the intervention had a positive effect. On average, coffee productivity in 2020 was `{r} round(100*(mean(data$coffee_prod)-mean(data$coffee_prod_past))/mean(data$coffee_prod_past), 1)`% greater than in 2015.
```{r}
#| eval: true
#| echo: false
#| warning: false
#| message: false
#| fig-width: 4
#| fig-height: 4.2
#| fig-align: center
#| fig-cap: "Changes in coffee productivity measured in 2015 and 2020 across 36 farms. Summary statistics shown on the right side of each boxplot show the mean ± 95% CI (n=36)."
#| label: fig-coffee-prod
library(Hmisc)
ggplot(data_bartlett, aes(x=factor(g), y=x))+
geom_boxplot(width=0.4, fill="#FFCD00")+
stat_summary(fun.data = "mean_cl_boot", position=position_nudge(x=0.4))+
theme_bw()+
xlab("")+
ylab("Coffee productivity (kg/ha/year)")+
theme(axis.text = element_text(color="black", size=12),
axis.title.y = element_text(margin=margin(r=10), size=13))
```
:::
::::